As you can see from the diagram below;
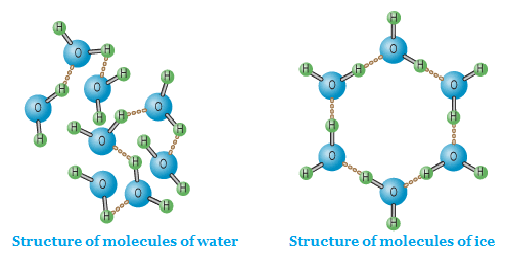
Ice molecules are loosely packed compare to water.
Ice is 9% less dense than water.
Ice is the solid state of water, a normal liquid substance that freezes to the solid state at temperatures of 0 °C (32 °F) or lower, and expands to the gaseous state at temperatures of 100 °C (212 °F) or higher.
Celsius (C) and Fahrenheit (F)
Ice is a form of water in which two elements Hydrogen (H) and Oxygen (O) are present in a fixed ratio of 2:1. These elements are in a chemically combined state. H2O.
Ice is a transparent, colorless substance with some special properties; it floats in water, ice expands when water freezes, and its melting point decreases with increase in temperature.
Moreover, ice weighs about 90% of its volume of water, therefore, 90% of the ice when placed in water, will be under water and the rest above.

Two glasses containing Water and Ice.

Ice cubes floating in a glass of water.